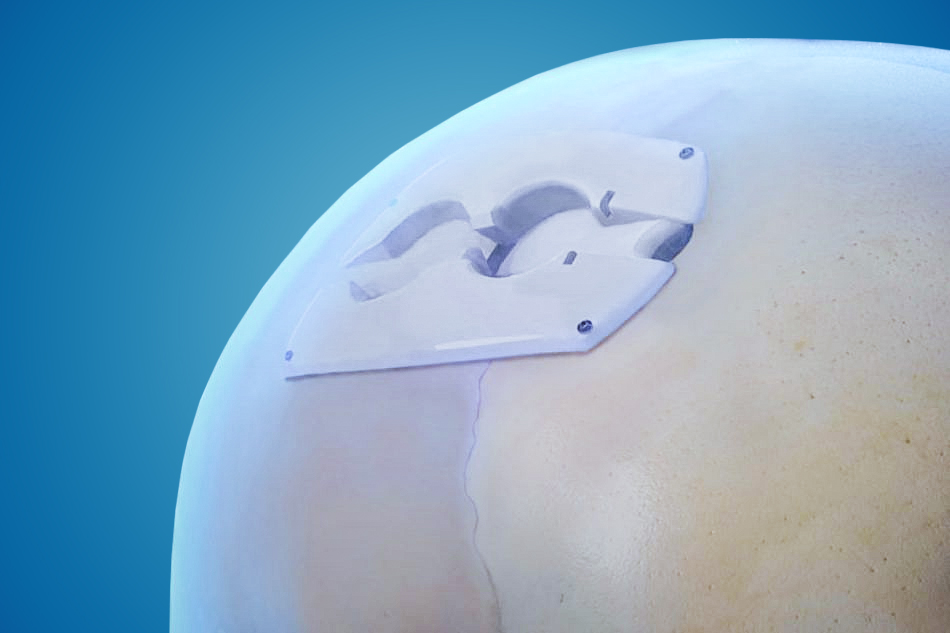
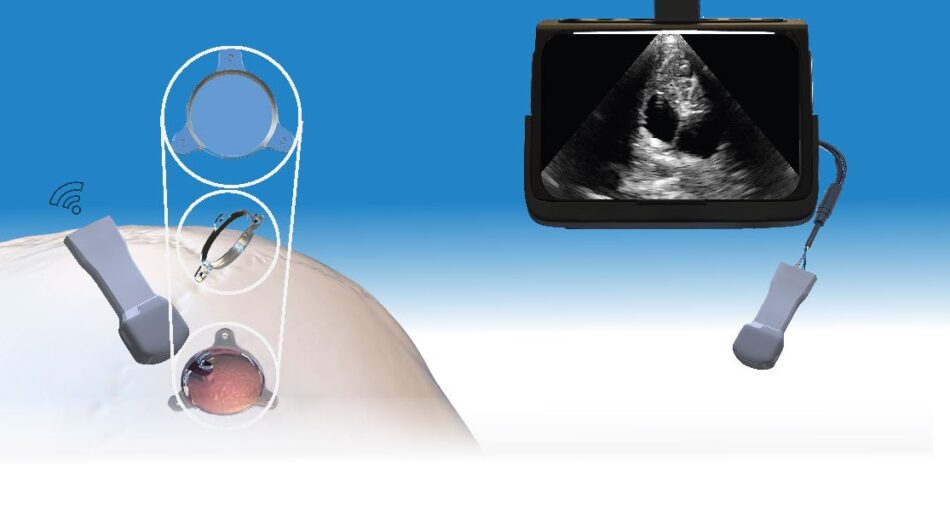
ClearFit® Cover
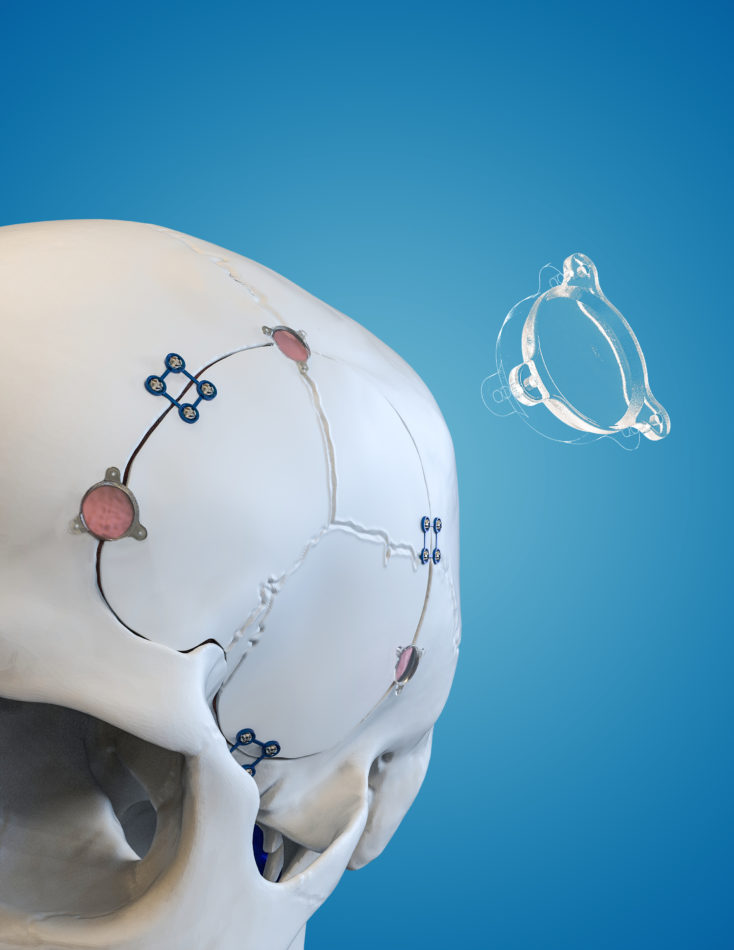
In 2016, in partnership with Johns Hopkins, Longeviti pioneered the development of the low-profile intracranial device platform – the world’s first customizable platform to address a growing unmet clinical need for patients undergoing complex brain surgeries.
Prior to Longeviti, there was no other solution that enabled the combination of technology within the cranium. Other solutions focus on only one of two primary neurological needs – either the functional requirements of the implant, or the reconstructive requirements. Longeviti solutions address both.
Since 2018, the company has brought several FDA-cleared implants to market. Longeviti’s technology is patent protected, view patents here.